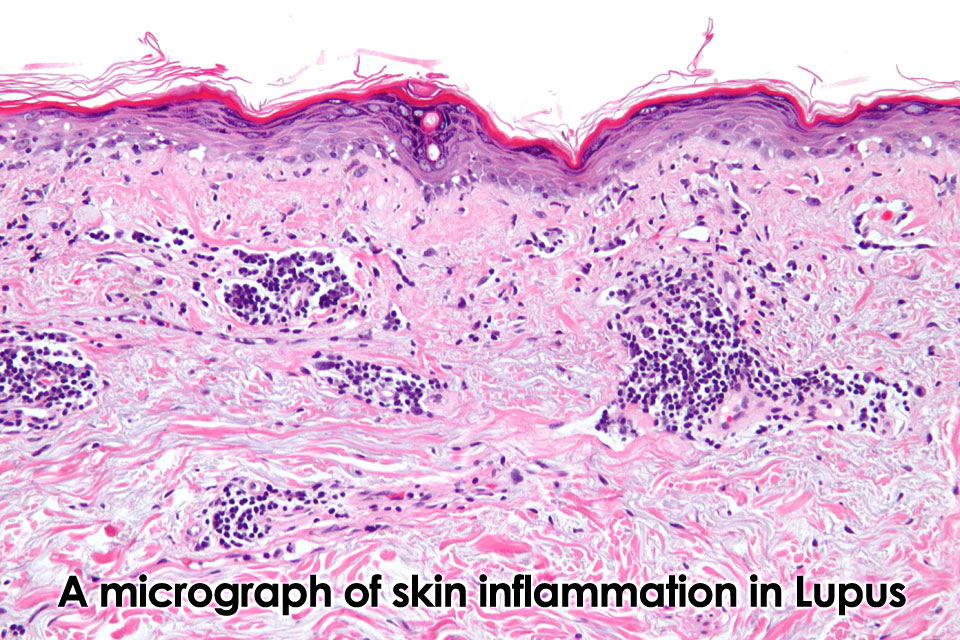
 An autoimmune disease is a disorder in which the immune system produces antibodies to an endogenous antigen, with the subsequent injury to the tissues of the body that display this antigen. Although the connective tissues like skin, muscles and joints are susceptible to wear and tear, some people have a tendency for these tissues to undergo degenerative changes that are initiated by inflammatory and immunological mechanisms. Inflammation of the skin and joints, such as systemic lupus erythematosus and rheumatoid arthritis, respectively, are among the most familiar forms of proinflammatory automimmune disorders, but other types of conditions have been found to have an autoimmune component, including Hashimoto’s thyroiditis, myasthenia gravis, diabtetes mellitus, and vitiligo. The modern medical approach of autoimmine disorders rests primarily upon the use of nonsteroidal antiinflammatory drugs (NSAIDs) and corticosteroids. While effective initially, it appears that many of these conditions respond poorly to these drugs over time, and with increased dosages there is an increased risk of iatrogenic effects.
An autoimmune disease is a disorder in which the immune system produces antibodies to an endogenous antigen, with the subsequent injury to the tissues of the body that display this antigen. Although the connective tissues like skin, muscles and joints are susceptible to wear and tear, some people have a tendency for these tissues to undergo degenerative changes that are initiated by inflammatory and immunological mechanisms. Inflammation of the skin and joints, such as systemic lupus erythematosus and rheumatoid arthritis, respectively, are among the most familiar forms of proinflammatory automimmune disorders, but other types of conditions have been found to have an autoimmune component, including Hashimoto’s thyroiditis, myasthenia gravis, diabtetes mellitus, and vitiligo. The modern medical approach of autoimmine disorders rests primarily upon the use of nonsteroidal antiinflammatory drugs (NSAIDs) and corticosteroids. While effective initially, it appears that many of these conditions respond poorly to these drugs over time, and with increased dosages there is an increased risk of iatrogenic effects.
Intestinal Permeability: Leaky Gut syndrome
There is an increasing amount of evidence that damage to the gut wall plays a role in autoimmune disease. A significantly high number of patients with ankylosing spondylitis, a rheumatoid-like condition of the axial skeleton and large joints, have been shown to have histological indications of chronic gastrointestinal inflammation and damage. It appears that in many of these patients the remission of the condition occurs in tandem with the remission of digestive inflammation, and vice versa (Mills and Bone 2000, 144-4).
In Ayurveda, most treatable disease arises from the presence of ama, a toxic by-product of poor digestion. One of the features of ama is that once it enters the dhatu cycle or tissues of the body, it serves as an obstructive element that impairs their nutrition and function. Ama has a particular affinity for tissues that are weak , such as old injuries, and tends to accumulate in these locations. Once it is firmly wedged in these locations, the doshas become vitiated: first kapha, with an increase in congestion; followed by pitta which sets up a cycle of inflammation; and then vata, which is responsible for the degenerative changes.
The theory of Intestinal Permeability derived in large part from the field of Functional Medicine, is that some agent or combination of agents initiates an inflammatory response in the digestive tract. Failure to remove this causes or causes, persistent GI inflammation eventually disrupts the integrity of the mucosal lining of the gut. The tiny perforations allow for molecules larger than usual to pass across the normally selectively permeable membrane of the gut, including molecules from dietary protein and fats, bacteria, parasites, and fungi. In response to this infiltration, an immune response is initiated and the body begins to manufacture specific antibodies against these antigens. Unfortunately, human tissues have antigenic markers that are almost identical to many of the substances that pass across a permeable intestinal wall. Once activated, these antibodies then circulate throughout the body to “look” for more antigens. When an antigen is found, such as a tissue that has similar markers to the exogenous antigen, the antibody initiates an immune response and the tissue begins inflamed, and over time, gradually degenerates. Apart from pre-existing digestive weakness or enzyme deficiencies (e.g. celiac disease, lactose intolerance), there are a number of factors that directly or indirectly promote gut irritation and inflammation, including:
- foods: anti-nutrient factors (cereals, legumes, nuts, seeds), enzyme inhibitors (cereals, legumes, nuts, seeds), lectins (cereals, legumes, nuts, seeds), alcohol, caffeine, refined carbohydrates/sugar, insufficient fiber intake, excess protein intake, Mallaird reactions (i.e. the “browning” reaction in proteins and carbohydrates from the application of heat), lipid peroxides (in refined/heated fats and oils), food preservatives, food additives, microparticles (e.g. flour products, titanium dioxide)
- medications: antibiotics, tylenol, NSAIDs, corticosteroids, oral contraceptives
- pathogens: parasites, pathogenic bacteria, fungal mycotoxins
Chronic gut inflammation damages the protective coating of IgA, the antibody that functions to maintain the integrity of the mucous membranes. As a result, the inhibition of a localized immune response allows for a decline of non-specific resistance, and an increased risk of viral, bacterial, fungal and parasite infection. The toxins that are produced by these microorganisms can inhibit liver function, and promote a heightened sensitivity to antigenic triggers such as cigarette smoke and strong perfumes. Thus many environmental sensitivities such as hayfever, can be caused or provoked by intestinal permeability.
Chronic gut inflammation also damages the absorptive capacity of the epithelium, and thus plays an important role in nutrient deficiency. Antibody-mediated destruction of transport proteins will also inhibit the absorption of these nutrients. Conditions such as osteoporosis and fibromyalgia that often display a deficiency of certain minerals such as zinc, calcium, and magnesium, can be reasonably linked with impaired absorption. Beyond minerals, similar deficiencies of vitamins, amino acids and essential fatty acids can lead the development of many different conditions, as well as generalized complaints as fatigue, irritability, poor concentration and headaches.
Achlorhydria (absence of stomach acid) and hypochlorhydria (deficiency of stomach acid) are increasingly recognized as a factor in leaky gut syndrome and autoimmune disorders, as well as many nutrient deficiencies such as Vitamin B12 (Kelly 1997). As we age, the parietal cells that secrete hydrochloric acid (HCl) begin to atrophy, and the levels of HCl drop. HCl is absolutely necessary for the activation of an enzyme called intrinsic factor, which assists in the absorption of vitamin B12, or extrinsic factor, necessary for proper nervous function and the formation of red blood cells. Other common nutrient deficiencies in hypochlorhydria include protein deficiencies, as well as mineral deficiencies such as chromium, copper, iron, magnesium, manganese, molybdenum, selenium, and zinc (Bergner 1997, 292). Apart from aging, the causes of low stomach acid include hereditary factors, chronic stress, and most commonly, the use of antacids and antiulcer medications such as cimetidine and ranitidine. Low stomach acid is difficult to diagnose, and many different techniques exist that can provide some indication. Hypochlorhydria can be inferred by the presence of pernicious anemia, poor gastric motility and poor digestion, determined as well from stool samples in which animal proteins will be poorly digested. Low stomach acid is best treated by dealing with chronic stress issues, using stimulants and carminatives to “warm” the stomach (e.g. ginger, cayenne, bayberry), followed later once appetite is restored with bitter herbs such as Gentian (Gentiana luteum) and Barberry (Berberis vulgaris) that move the liver and promote peristalsis. Broad spectrum digestive enzymes that include HCl, papain, bromelain, pancreatin, and lipase may be helpful, particularly in the aged. Additional measures include the consumption of sour-tasting remedies and foods before and with meals, such as apple cider vinegar or live culture vegetable pickles.
Another key factor in the equation is the gastrointestinal ecology, including the presence of probiotic organisms such as Lactobacillus cidophilus and Bifidobacterium bifidum. Research has shown that these friendly bacteria enhance the capacity of the immune cells in the gastrointestinal tract to defend against foreign pathogens. These immune cells maintain an important link to other cells in the immune system, and appear to have a modulating role in immune function. Clinical research has shown that the oral supplementation of friendly bacteria, particularly in the form of traditional live-culture foods (e.g. sauerkraut), enhances phagocytic activity and provides sustained immunological protection, both within and without the gastrointestinal tract. Additionally, these bacterium compete with other pathogenic organisms such as Candida albicans and the diarrhea-causing Clostridium difficile.
Treatment of Autoimmune disorders
The treatment of autoimmune disorders is strengthened by an understanding of the underlying energetic principle of treatment. Using an Ayurvedic approach, autoimmune conditions with a kapha-type variant would benefit from warm, dry and stimulating therapies, to remove congestion, enhance circulation and promote digestion. Pitta-type variants of autoimmune disorders benefit from cooling ,moistening and grounding therapies. Vata-type variants of autoimmune disorders require warming, moistening and rejuvenating therapies.
Generally, the treatment of autoimmune conditions involves several steps, each of which is typically applied in succession, but can be adapted as needed:
1. Restore proper digestion
According to Ayurveda, the fundamental cause of autoimmune disease relates to the disease called amavata. In this condition, ama accumulates due to weak digestion, where over time it gradually establishes an entropic cycle of decay and degeneration (i.e. vata). The primary method to remove ama is by the enhancement of digestion. This is done by gentle fasting, using light and easily digestible foods, and by the use of digestive stimulants and carminatives. Examples of helpful foods include warm water, vegetable soup, rice soup, kitchari, meat soup, and bone broth. Examples of useful herbs include Ginger (Zingiber officinalis), Pippali (Piper longum), Cayenne (Capsicum spp.), Bayberry (Myrica cerifera), Ajwain (Trachyspermum ammi), Cardamom (Elettaria cardamomum), and Cinnamon (Cinnamomum zeylanica).
2. Support liver function and enhance detoxification.
The notion of modulating liver function is not an area that is given much consideration in modern medicine, probably because the rationale for such an approach is derived from older ideas on the subject of autotoxicity, a concept routinely disregarded by self-acclaimed rationalists. The importance of the liver in maintaining health cannot be argued, however, and with increasing exposure to environmental contaminants, mutagens and xenobiotics in modern society, liver detoxification pathways can become quickly overwhelmed. Fortunately, there are many botanicals that support liver detoxification pathways, and also demonstrate hepatoprotective and hepatoregenerative properties. Considering the important of the liver in digestive function, herbs that support liver function shouldbe included in any protocol for autoimmune conditions. These include Barberry (Berberis spp.), Turmeric (Curcuma longa), Licorice (Glycyrrhiza glabra), Boldo (Peumus boldo), Bhumyamalaki (Phyllanthus amarus), Katuka (Picrorrhiza kuroa), Dan Shen (Salvia miltiorrhiza), Wu Wei Zi (Schizandra chinensis), Huang Qin (Scutellaria baicalensis), Milk Thistle (Silybum marianum), and Guduchi (Tinospora cordifolia). Additional botanicals that have an affinity for removing congestion and eliminating wastes generally from the body include Celery seed (Apium graveolens), Burdock (Arctium lappa), Cleavers (Galium aparine), Red Clover (Trifolium pratense), Nettle (Urtica dioica), and the formula Triphala.
3. Reduce and extinguish the inflammatory cascade.
The role of inflammation is the chief concern of modern medicine when it comes to the treatment of autoimmune disorders. Unfortunately, patients with autoimmune disorders are in a weakened state, and the usual approach consisting of methods to suppress inflammation, including NSAIDs (which promote liver damage and irritate the gut wall) and corticosteroids (which depress immune function) do little to resolve the issue, and make things worse over the long run. What is missing in medicine is a definitive understanding of the etiology of autoimmunity. In part this is due to a myopic perspective on specific molecular mechanisms or certain genes that are active in autoimmunity, and not an examination of the broader issue. As I describe in my book Food As Medicine, autoimmunity is inherently linked to the advent of agriculture and the consumption of agricultural staples. Removing these foods from the diet is thus an important way to reduce inflammation and autoimmunity, by addressing the cause itself. To this end, I typically suggest a Paleolithic diet – a measure have successfully used in countless cases over the last 20 years.
Given the inherent issue of using immunosuppressant drugs, botanicals with anti-inflammatory properties are a much safer alternative, but must be applied only after digestion has been established. In Ayurveda, this relates to the use of herbs which reduce pitta, and hence are associated with the liver and biliary function. Many anti-inflammatories are thus cholagogues, upregulating and supporting hepatic detoxification, stimulating the excretion of bile. Anti-inflammatory botanicals include: Turmeric (Curcuma longa), Amla berry (Emblica officinalis), Licorice root (Glycyrrhiza glabra), Lignum vitae (Guaiacum officinalis), Devil’s Claw (Harpagophytum procumbens), Buckbean (Menyanthes trifolata), Trembling Aspen (Populus tremuloides), Willow (Salix spp.), Huang Qin (Scutellaria baicalensis), Sarsaparilla (Smilax spp.), Feverfew (Tanacetum parthenium), Ashvagandha (Withania somnifera), Black Cohosh (Cimicifuga racemosa), and Wild Yam (Dioscorea villosa)
If there is mucosal inflammation and/or bleeding, even before digestive stimulants can be used, the initial use of demulcents and astringents may be considered. Generally, demulcents are helpful to reestablish the integrity of the epithelial lining of the gut, and are better for constipative conditions. Astringents tone the mucous membranes and firm the musculature of the bowel, and are more appropriate for diarrheal conditions. Inflammation an also result in spasm, and hence antispasmodics can have a role to play to relieve abdominal pain and hyperperistalsis, slowing the frequency of loose motions. Of special note is the potentially toxic Belladonna (Atropa belladonna), which through the antimuscarinic activity of its tropane alkaloids inhibits peristalsis and gastrointestinal hypersecretion. Some analgesic herbs also exhibit this effect:
- Demulcents: Kumari (Aloe vera juice), Marshmallow root (Althaea officinalis), Slippery Elm (Ulmus fulva), Licorice root (Glycyrrhiza glabra)
- Antispasmodics: Black Cohosh (Cimicifuga racemosa), Wild Yam (Dioscorea villosa), Belladonna (Atropa belladonna- TOXIC)
- Astringents: Bayberry (Myrcia cerifera), White Oak (Quercus alba), Goldenseal (Hydrastis canadensis), Witch Hazel (Hamamelis virginiana), Sanqi (Panax notoginseng – a hemostatic that also restores proper circulation)
- Analgesics: Wild Lettuce (Lactuca virosa), Jamaican Dogwood (Piscidia erythrina), Nutmeg (Myristica fragrans) and California Poppy (Eschscholzia californica), Opium Poppy (Papaver somniferum)
4. Address dysbiosis and support immune function.
One driving mechanism behind autoimmunity is infection from pathogenic viruses, bacteria, fungi, or parasites. When this ecological imbalance prevails the general term to describe it is dysbiosis – in essence – a “wrong” of life. While digestion is a key element in the restoration of ecological balance, “population control” measures sometimes need to be undertaken to restore balance. This includes the use of antivirals, antimicrobials, and botanicals that either stimulate or modulate immune function. The use of herbs with an immunostimulant activity in autoimmune conditions is something of a controversy among practitioners, because if an autoimmune condition is an expression of an immune system gone out of control, stimulation could exacerbate the condition. In part this argument is limited by semantics, as plants and their activities do not fit into neat little boxes. Nonetheless, the excessive or isolated use of directly immunostimulant botanicals such as Echinacea should be approached with caution in autoimmune disorders. Another category of botanicals called immunomodulants are perhaps more appropriate to autoimmune conditions, although where one herb ceases being immunostimulant and becomes immunomodulant is a gray area, and is likely highly individual.
- Antivirals: Garlic (Allium sativum), St. John’s Wort (Hypericum perforatum), Ban Lan Gen (Isatis tinctoria), Biscuit root (Lomatium dissectum), Osha (Ligusticum spp.), Lemon Balm (Melissa officinalis), Cubeb (Piper cubeba), and Western Red Cedar (Thuja plicata).
- Antibacterials: Wild Indigo (Baptisia tinctoria), Guggulu (Commiphora mukul), Purple Coneflower (Echinacea angustifolia), Huang Lian (Coptis chinense), Lian Qiao (Forsythia suspens), Jin Yin Hua (Lonicera japonica), Ban Lan Gen (Isatis tinctoria), Huang Qin (Scutellaria baicalensis), and Goldenseal (Hydrastis canadensis).
- Paracidals: Garlic (Allium sativum), Sweet Annie (Artemisia annua), Nimba (Azadirachta indica), Malefern (Dryopteris felix-mas), and Kutaj (Holarrhena antidysenterica)
- Fungicidals: Sweet Annie (Artemisia annua), Neem (Azadirachta indica), Huang Lian (Coptis chinense), Bhringaraj (Eclipta alba), Toothache plant (Spilanthes acmella), Pau d’Arco (Tabebuia spp.), Haritaki (Terminalia chebula), and Vibhitaki (Terminalia bellerica).
- Immunostimulants: Red root (Ceanothus spp.), Purple Coneflower (Echinacea angustifolia), Biscuit root (Lomatium dissectum), and Western Red Cedar (Thuja plicata)
- Immunomodulants: Huang Qi (Astragalus membranaceus), Amalaki (Emblica officinalis), Ling Zhi (Ganoderma spp.), Siberian Ginseng (Eleuthrococcus senticosus), Wu Wei Zi (Schizandra chinense), and Ashvagandha (Withania somnifera).
5. Adjust flora of the bowel.
Once digestion has been established, and after the use of paracidals, antibacterials, and antifungals to address the issue of dysbiosis, measures should be taken to support a healthy gut flora, including the use of both prebiotics and probiotics. Prebiotics include polysaccharides, including our own mucosal secretions – which is why digestive stimulants are an important aspect of treatment. Recent research has suggested that the polysaccharides produced by the mucosa acts as a first line of defense against pathogens by binding with carbohydrate-binding proteins on bacterial membranes. This “decoy” activity disables the bacteria from adhering to the mucosa of the gut wall. In a similar fashion, polysaccharides from exogenous sources exhibit the same activity – including medicinal herbs. Further, some polysaccharides such as inulin appear to be almost exclusively fermented by bifidobacteria, the so-called “friendly” bacteria (Roberfroid 1993). The use of inulin leads to an overall increase in fecal bacterial biomass, a decrease in ceco-colonic pH, and a positive effect upon lipid metabolism. Many medicinal plants contain inulin, such as Burdock (Arctium lappa) and Dandelion root (Taraxacum officinale).
Probiotics refer to the oral consumption of “friendly” bacteria that helps to restore a depleted or dysbiotic gastrointestinal microbiome. This includes species such as Lactobacillus acidophilus and Bifidobacterium bifidum, and also specialized yeasts such as Saccharomyces boulardii. The most sustainable choice for probiotics are traditional live-culture fermented foods, such as sauerkraut and other lacto-fermented vegetables. Yeasted or foods prepared with a SCOBY such as kefir and kombucha are not advised. Supplements are often recommended, but there is good reason to be skeptical that they have any viability after manufacturing, and should only be relied upon when food sources aren’t being applied.
6. Eliminate or reduce the insult to the gut barrier.
Diet is an all important factor in autoimmune disease, and care must be taken to eliminate those foods which initiate an immune response. To determine which foods may be a factor in the disorder, an eliminative diet is best undertaken, removing the most common of food allergens from the diet for a period of two weeks, and then gradually introducing them back into the diet, one at a time over a period of one week, and noting any symptoms. Other important strategies to enhance gut function include:
- Limiting the use of antibiotics and NSAIDS as they can cause gastrointestinal irritation. Even though it is not known exactly known how NSAIDs cause damage to the gut, several hypotheses have been suggested, including enhanced intestinal permeability, inhibition of cyclooxygenase, enterohepatic recirculation, and formation of adducts (Cipolla et al 2002).
- Reducing exposure to xenobiotics such as pesticides, insecticides and herbicides, emphasizing organically grown vegetables and free range, hormone/antibiotic-free animal produce.
- Increasing intake of high fiber foods and foods rich in antioxidant phytochemicals such as broccoli, cabbage, cauliflower, beets, carrots and onions.
- Increasing intake of omega 3 fatty acids, such as salmon, halibut and arctic char or supplementing with essential fatty acids high in omega 3, such as flax and hemp oil, to inhibit the inflammatory cascade.
- Supplementing with a chelated multimineral and trace mineral supplement to provide for the manufacture of detoxification enzymes and antioxidants.
- Supplementing with digestive enzymes (e.g. bromelain, HCl, pancreatin) to counter hypochlorhydria and to improve digestion. Bromelain and pancreatin also inhibit antiinflammatory prostaglandins and leukotrienes.
- Supplementing with glucosamine to improve bowel wall integrity
- Supplementing with antioxidants such as N-acetyl cysteine to limit oxidative stress, but only after protozoal infections have been dealt with.
- Vitamin D3, to modulate the immune response: 3000-10,000 IU/d.
To accurately assess for intestinal permeability, diagnostic laboratories now offer a convenient method of testing. This simple test measures the ability of two non-metabolized sugar molecules, mannitol and lactulose, to permeate the intestinal mucosa. Mannitol is easily absorbed and serves as a marker of transcellular uptake, while lactulose is only slightly absorbed and serves as a marker for mucosal integrity. To perform the test, the patient mixes pre-measured amounts of lactulose and mannitol and drinks the challenge substance. The test measures the amount of lactulose and mannitol recovered in a urine sample over the next 6 hours.